How HEOR Frameworks Are Reshaping Modern Healthcare Decision-Making Globally
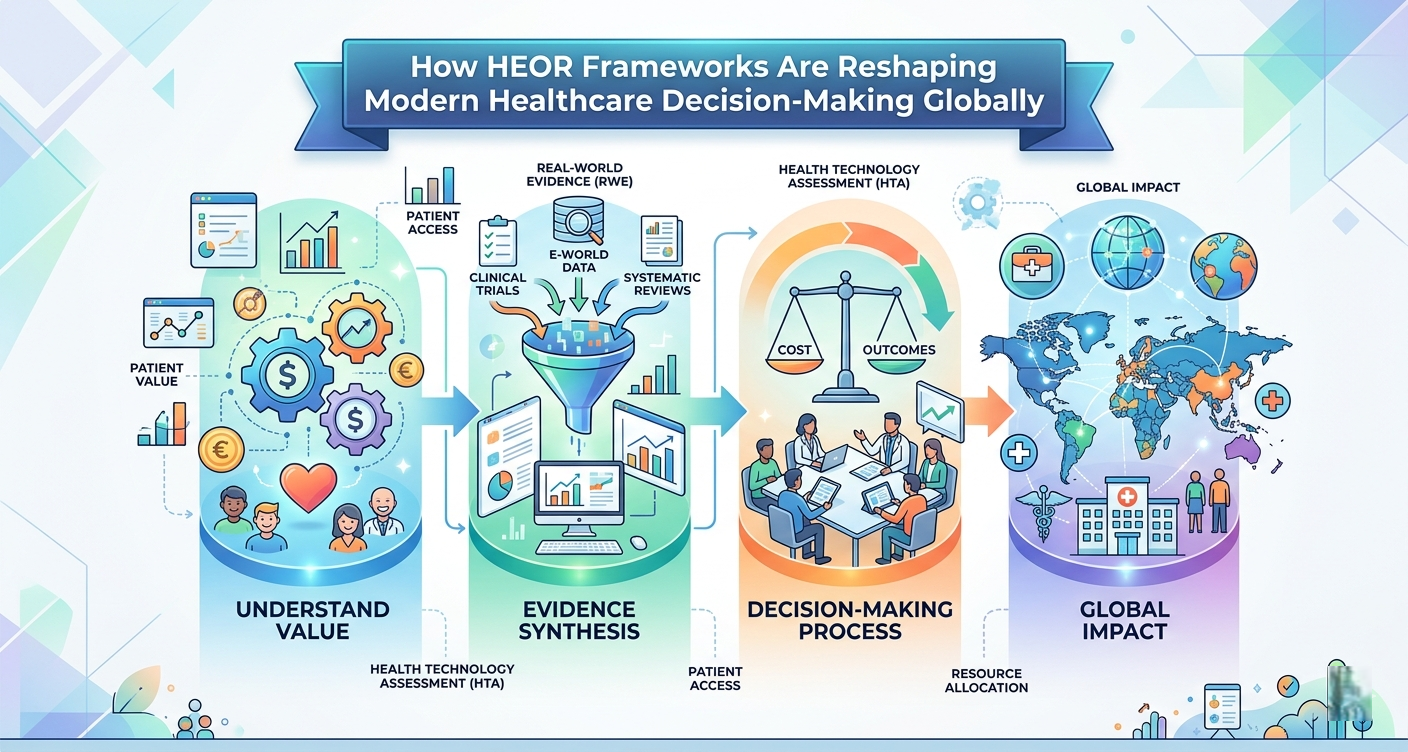
In today's rapidly evolving healthcare landscape, evidence-based decision-making has become the cornerstone of sustainable health systems. Payers, regulators, and pharmaceutical companies are increasingly turning to Health Economics and Outcomes Research (HEOR) to justify clinical investments, support market access, and ultimately improve patient outcomes. From understanding disease burden to navigating complex reimbursement landscapes, HEOR methodologies provide the analytical backbone that modern healthcare cannot afford to ignore.
The Role of Survival Analysis in Outcomes Research
One of the most critical components in evaluating how patients respond to therapies over time is survival analysis healthcare outcomes. This statistical approach models the time until an event of interest — such as disease progression, hospitalization, or death — occurs within a patient population. By examining survival curves and hazard ratios, researchers can draw meaningful comparisons between treatment arms, offering stakeholders a quantitative lens through which long-term clinical value is assessed.
Survival analysis is particularly indispensable in oncology and chronic disease research, where understanding mortality reduction and progression-free intervals directly influences formulary decisions and clinical guidelines. Without this rigorous methodology, economic models would rest on incomplete foundations, undermining the credibility of any downstream evaluation.
Mapping Clinical Journeys Through Treatment Pathway Analysis
Beyond survival data, understanding how patients actually move through the healthcare system is equally vital. Treatment pathway analysis HEOR examines the sequence of therapeutic interventions a patient receives from diagnosis through disease management. This form of real-world evidence generation helps identify gaps in care, treatment switching patterns, and areas where clinical practice diverges from established guidelines.
For pharmaceutical manufacturers and health systems alike, mapping these pathways reveals where a new intervention might fit within the existing standard of care. It also uncovers opportunities to demonstrate superiority or cost-effectiveness versus current treatments — a critical data point when approaching payers with a value proposition.
Bridging Clinical Evidence and Economic Value
The intersection of clinical outcomes and economics defines the very essence of HEOR. HTA economic evaluation encompasses a range of techniques — including cost-effectiveness analysis, cost-utility analysis, and budget impact modeling — designed to quantify the economic consequences of adopting a new health technology relative to existing alternatives. These evaluations translate clinical trial endpoints into economic metrics such as cost per quality-adjusted life year (QALY), making the value of therapies legible to decision-makers who must allocate limited resources wisely.
A well-constructed economic evaluation not only strengthens a market access submission but also builds a compelling narrative around patient benefit. When healthcare systems operate under fiscal pressure, the ability to demonstrate measurable value becomes a competitive differentiator for any therapy seeking formulary inclusion.
Navigating the HTA Landscape for Market Access
Regulatory approval alone no longer guarantees patient access. Products must also satisfy the rigorous scrutiny of national health technology bodies such as NICE in the UK, G-BA in Germany, and HAS in France. Health technology assessment analysis provides the structured framework through which these agencies evaluate comparative effectiveness, safety, and economic value before recommending reimbursement.
Preparing a robust HTA submission demands early planning, cross-functional collaboration, and a deep understanding of the evidentiary standards expected by each jurisdiction. Companies that invest in HTA-aligned evidence generation from the earliest stages of clinical development are better positioned to achieve favorable outcomes when the time for market access negotiations arrives.
Building Winning Reimbursement Strategies
Even the most clinically innovative product can fail commercially without a coherent, data-driven approach to payer engagement. Reimbursement strategy analysis HEOR integrates epidemiological data, payer landscape assessments, and economic modeling to craft a strategy that anticipates payer objections and pre-emptively addresses them with evidence. This includes identifying the right comparators, selecting appropriate outcomes measures, and aligning clinical messaging with the value frameworks payers use.
A sophisticated reimbursement strategy is not developed in isolation — it draws directly from survival analyses, treatment pathway insights, and HTA requirements, weaving them into a unified narrative that resonates with diverse stakeholder audiences across geographies.
Conclusion
HEOR has evolved from a niche discipline into an indispensable pillar of modern healthcare strategy. Whether through survival analysis healthcare outcomes, treatment pathway analysis HEOR, HTA economic evaluation, health technology assessment analysis, or reimbursement strategy analysis HEOR, organizations that embed these methodologies early in their development lifecycle are far better equipped to achieve market access, demonstrate patient value, and sustain long-term commercial success in increasingly competitive global healthcare markets.