The Strategic Evolution of the Global Pharmacovigilance Outsourcing Market
The global healthcare landscape is undergoing a massive transformation as pharmaceutical companies face increasingly complex regulatory environments and a rising volume of adverse drug event data. This shift has propelled the Global Pharmacovigilance Outsourcing Market into a period of rapid growth. Pharmaceutical and biotechnology firms are moving away from in-house safety management, choosing instead to partner with specialized service providers to streamline their drug safety operations and ensure global compliance.
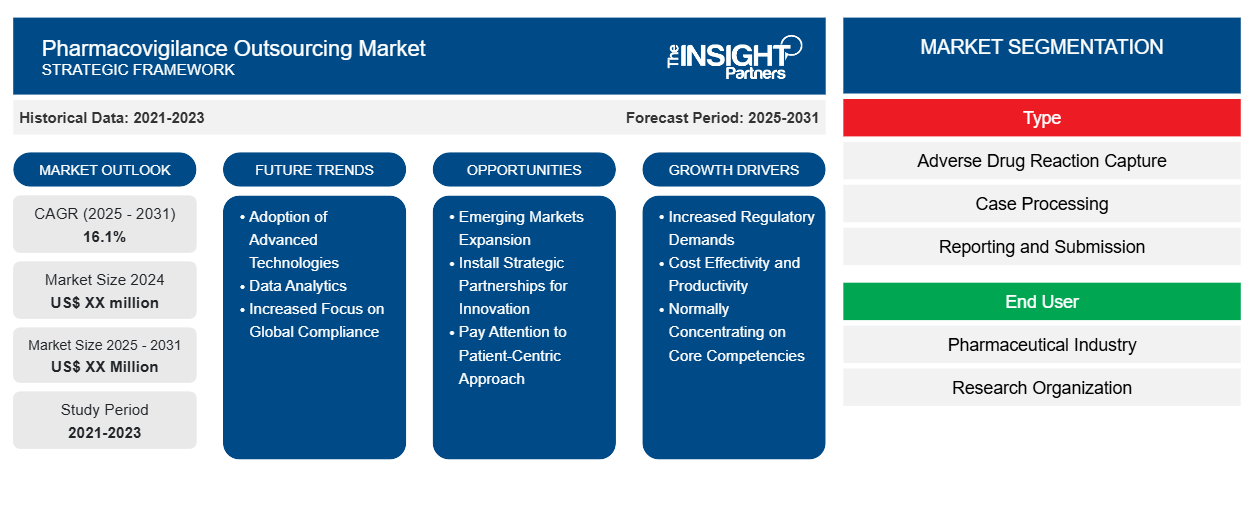
According to the latest market analysis, the Global Pharmacovigilance Outsourcing Market is expected to witness significant growth in the coming years. Based on insights from The Insight Partners, the broader pharmacovigilance market is estimated to register a Compound Annual through 2031. Furthermore, current industry projections indicate that the specialized outsourcing segment is expected to register an even more aggressive CAGR of 16.1% from 2025 to 2033, reflecting the urgent shift toward partner-led safety models.
Download Sample PDF : https://www.theinsightpartners.com/sample/TIPRE00040657
Market Drivers: What is Fueling the Outsourcing Surge?
The expansion of the pharmacovigilance outsourcing sector is fueled by several critical industry dynamics:
- Rising Incidence of Adverse Drug Reactions (ADRs): The increasing global consumption of medicines, coupled with the prevalence of chronic diseases, has led to a surge in reported adverse events. As the volume of safety data grows beyond the capacity of internal departments, outsourcing to Contract Research Organizations (CROs) and Business Process Outsourcing (BPOs) becomes essential for efficient data processing and signal detection.
- Stringent Regulatory Requirements: Regulatory bodies worldwide are continuously updating safety guidelines to prioritize patient safety. Compliance now requires 24/7 monitoring, real-time reporting, and sophisticated risk management plans. Outsourcing partners provide the expertise and infrastructure necessary to meet these rigorous standards without the heavy capital investment required for internal upgrades.
- Digital Transformation and AI Integration: The integration of Artificial Intelligence (AI) and Machine Learning (ML) in drug safety is a major trend. Leading outsourcing providers are deploying automated case processing and advanced analytics to improve the accuracy of signal detection. This technological edge allows pharmaceutical companies to access cutting-edge tools through their partners, driving market demand.
- Focus on Core Competencies: By outsourcing the resource-intensive tasks of data entry and clinical trial safety monitoring, pharmaceutical giants can refocus their internal efforts on Research and Development (R&D) and core business strategies. This operational efficiency is a significant driver for both large-scale manufacturers and emerging biotech firms.
High-Growth Opportunities for Stakeholders
As we look toward the 2026 to 2031 period, several untapped opportunities are emerging within the pharmacovigilance outsourcing space:
1. Expansion into Emerging Markets While North America remains the largest market, the Asia Pacific region offers the most significant growth opportunities. Countries like India and China have become global hubs for pharmacovigilance outsourcing due to lower operational costs, a large pool of skilled healthcare professionals, and an increasing number of clinical trials.
2. Precision Pharmacovigilance for Biologics The rise of biologics, biosimilars, and orphan drugs presents a specialized niche. These complex therapies require more granular safety monitoring and long-term surveillance. Service providers who develop specialized expertise in these therapeutic areas can capture a higher market share.
3. Real-World Evidence (RWE) Integration There is a growing opportunity for outsourcing partners to move beyond traditional case processing and into the realm of Real-World Evidence. By leveraging data from electronic health records (EHRs) and patient registries, providers can offer predictive safety insights that help pharma companies mitigate risks long before they lead to product recalls.
Top Market Players
The competitive landscape is characterized by a mix of specialized safety vendors and global CROs. Some of the top players shaping the industry include:
- Accenture
- Cognizant
- ICON plc
- Labcorp Drug Development (Covance)
- IBM Corporation
- ArisGlobal
- Capgemini (IGATE Corporation)
- Parexel International Corporation
- Wipro Limited
Related Report : Pharmacovigilance and Drug Safety Software Market Key Players and Opportunities by 2031
Future Outlook
The future of the hyaluronidase market looks promising as pharmaceutical companies continue to explore new therapeutic applications for the enzyme. With increasing adoption in biologic drug delivery, oncology treatments, and cosmetic procedures, the market is expected to expand significantly over the next decade.
Additionally, ongoing research in recombinant enzyme technology and personalized medicine is likely to create new opportunities for hyaluronidase applications. As healthcare systems worldwide focus on improving treatment efficiency and patient outcomes, hyaluronidase will continue to play an important role in modern medicine.
About Us
The Insight Partners is a global market research and consulting firm dedicated to delivering high-quality industry insights and strategic intelligence. The company provides comprehensive market reports covering technology, healthcare, manufacturing, and other major industries. Its research helps businesses understand market trends, identify growth opportunities, and make data-driven decisions
Contact Us
The Insight Partners
Phone: +1-646-491-9876
Email: sales@theinsightpartners.com
Website: https://www.theinsightpartners.com






