Virtual Clinical Trials Market 2034: US Leads Global Growth with Digital Transformation in Clinical Research
The Virtual Clinical Trials (VCTs), also known as decentralized clinical trials, are transforming the global clinical research landscape by leveraging digital technologies, remote patient monitoring, telemedicine, wearable devices, and cloud-based data platforms. These trials reduce the need for physical site visits, enabling patients to participate from home while maintaining data accuracy, regulatory compliance, and operational efficiency. The model enhances patient recruitment, improves retention rates, and accelerates drug development timelines across therapeutic areas.
Virtual Clinical Trials Market Size, Growth, and Forecast 2034
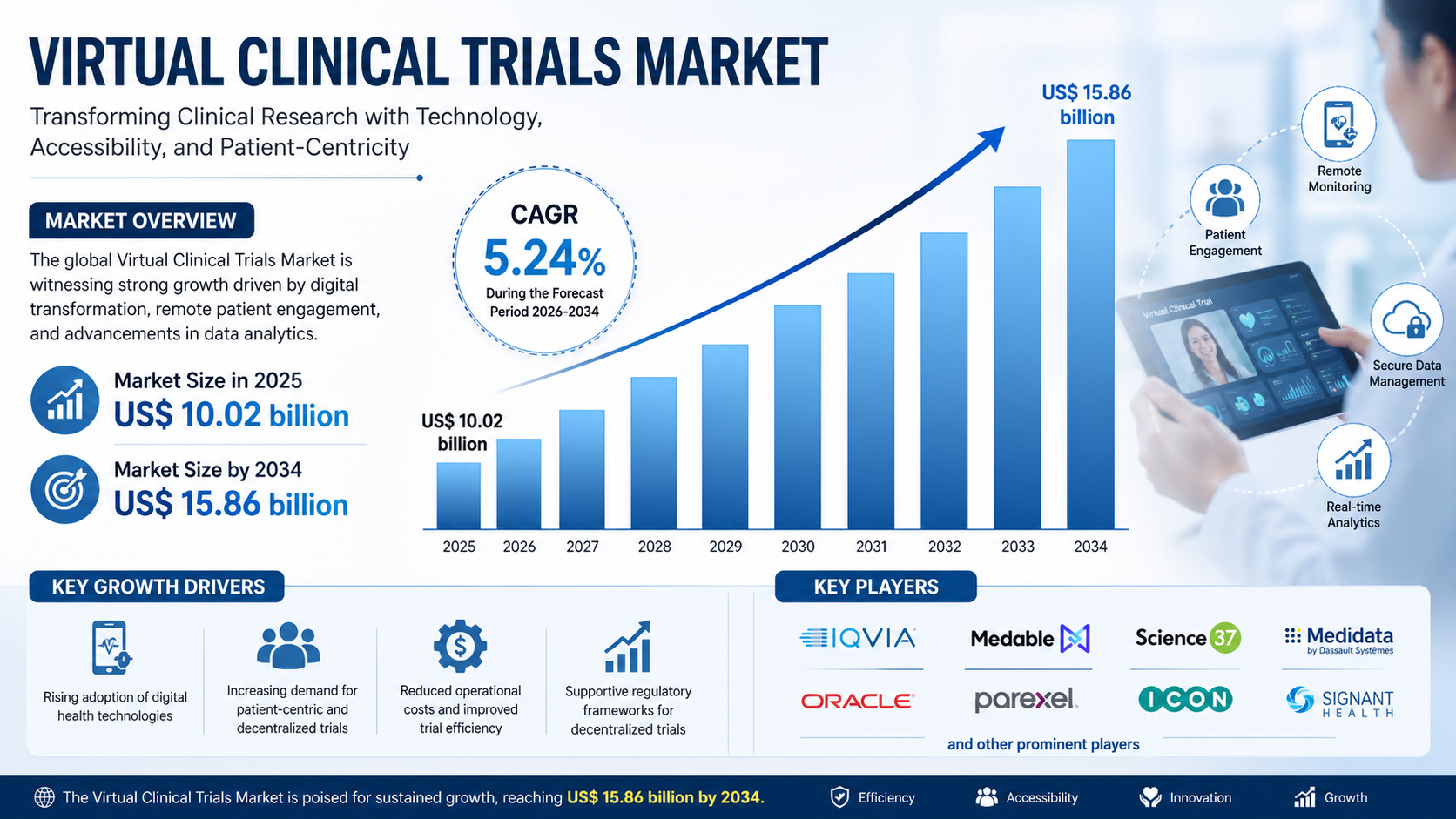
The global virtual clinical trials market size is projected to reach US$ 15.86 billion by 2034 from US$ 10.02 billion in 2025. The market is anticipated to register a CAGR of 5.24% during the forecast period 2026 to 2034. This steady growth reflects increasing investments in digital health infrastructure, rising clinical trial complexity, and the expansion of precision medicine initiatives.
Key Growth Drivers of the Virtual Clinical Trials Industry
The expansion of the virtual clinical trials market is driven by several critical factors. One of the primary drivers is the rising demand for faster drug development cycles. Traditional clinical trials often face delays due to patient recruitment challenges and geographic limitations. Virtual trials eliminate many of these barriers by enabling remote participation.
Another major driver is the increasing adoption of wearable health technologies and mobile health applications. These tools allow continuous monitoring of patient health data, improving trial accuracy and reducing reliance on in-person visits. Additionally, artificial intelligence and cloud computing are playing a significant role in streamlining data management and predictive analytics in clinical research.
Regulatory support is also contributing to market expansion. Agencies such as the FDA and EMA are encouraging decentralized trial models to enhance patient accessibility and data transparency.
US Leads the Virtual Clinical Trials Market Globally
The United States virtual clinical trials market due to its advanced healthcare infrastructure, strong presence of leading pharmaceutical companies, and early adoption of digital health technologies. The country has a well-established regulatory framework that supports decentralized clinical research, making it the preferred hub for clinical trial innovation.
High investment in biotechnology and life sciences research further strengthens the US market position. Leading pharmaceutical firms and contract research organizations in the country are increasingly integrating virtual trial platforms to improve operational efficiency and accelerate time-to-market for new therapies. Additionally, strong collaboration between technology providers and healthcare institutions is driving innovation in remote patient monitoring and data analytics solutions.
The growing prevalence of chronic diseases and the increasing demand for personalized medicine are also boosting clinical trial activity in the United States. As a result, the US is expected to maintain its leadership position in the global virtual clinical trials market throughout the forecast period.
Segmentation Analysis of Virtual Clinical Trials Market
The virtual clinical trials market can be segmented based on study type, phase, therapeutic area, and end user.
By study type, the market is divided into interventional studies and observational studies. Interventional studies dominate the segment due to their extensive use in drug development and regulatory approval processes.
By phase, the market includes Phase I, Phase II, Phase III, and Phase IV trials. Phase III trials hold the largest share as they involve large patient populations and require efficient data management systems.
By therapeutic area, oncology, cardiovascular diseases, neurology, and infectious diseases represent key segments. Oncology trials are particularly prominent due to the increasing demand for targeted cancer therapies and precision medicine approaches.
By end user, pharmaceutical and biotechnology companies lead the market, followed by contract research organizations and academic research institutes. Pharmaceutical companies rely heavily on virtual trials to reduce costs and improve patient engagement.
Get Sample PDF @ https://www.theinsightpartners.com/sample/TIPRE00015952
Top Players in the Virtual Clinical Trials Market
The virtual clinical trials market is highly competitive, with several global players investing in digital trial technologies and decentralized research platforms. Key companies include:
- Covance Inc
- CRF Health
- LEO Innovation Lab
- Clinical Ink, Inc.
- Paraxel International
- Medable, Inc
- Oracle Corporation
- Science 37
- ICON plc
- Medidata Solutions, Inc.
These companies are focusing on strategic partnerships, platform innovation, and artificial intelligence integration to enhance clinical trial efficiency and patient experience.
Regional Analysis of Virtual Clinical Trials Market
North America holds the largest share of the global virtual clinical trials market, driven by strong technological infrastructure and high adoption of digital health solutions. The United States, in particular, is the primary contributor to regional growth.
Europe follows closely, supported by favorable regulatory policies and increasing investments in healthcare digitization. Countries such as Germany, the United Kingdom, and France are actively adopting decentralized trial models.
The Asia Pacific region is expected to witness the fastest growth during the forecast period. Rising healthcare investments, growing clinical research activities, and expanding pharmaceutical manufacturing in countries like China, India, and Japan are key growth drivers.
Latin America and the Middle East and Africa are also emerging markets, with increasing awareness of virtual clinical trials and improving healthcare infrastructure supporting gradual adoption.
Future Outlook of the Virtual Clinical Trials Market 2034
The future of the virtual clinical trials market is expected to be shaped by continued advancements in digital health technologies, artificial intelligence, and real-world evidence integration. The adoption of hybrid trial models combining virtual and traditional approaches will become more common, offering greater flexibility and efficiency.
About The Insight Partners
The Insight Partners delivers market intelligence and consulting services to help clients make informed decisions. The firm covers industries such as Aerospace and Defense, Automotive and Transportation, Semiconductor and Electronics, Biotechnology, Healthcare IT, Manufacturing, Medical Devices, Technology, Media, and Chemicals and Materials.
Contact Us
Email: sales@theinsightpartners.com
Website: www.theinsightpartners.com
Phone: +1-646-491-9876
Also Available in :