Pharmaqo Labs: Standards, Progress, and the Modern Pharmaceutical Landscape

Introduction
The pharmaceutical industry today operates in a highly advanced and competitive environment where expectations are significantly higher than in the past. Consumers are more informed, regulatory standards are stricter, and manufacturing technologies are far more sophisticated. In this setting, companies must prioritize quality, consistency, transparency, and innovation to remain relevant. Within discussions about modern pharmaceutical manufacturing, Pharmaqo Labs is often referenced in relation to structured production systems and evolving industry practices.
The shift in consumer behavior has been one of the most influential changes in the industry. People now actively research products, compare quality standards, and evaluate company credibility before making decisions. This has forced manufacturers to adopt more disciplined approaches to production and customer communication.
Transformation of the Pharmaceutical Industry
Over time, the pharmaceutical industry has shifted from basic mass production to highly controlled, research-driven manufacturing. This transformation has been driven by technological advancement, global competition, and rising consumer awareness.
Modern pharmaceutical companies are no longer judged only by output but also by the integrity of their processes. Customers expect safety, accuracy, and reliability at every stage of production. As a result, companies are increasingly investing in advanced systems that ensure consistency and compliance with industry expectations.
In this evolving environment, Pharmaqo Labs is frequently mentioned in discussions about how modern pharmaceutical businesses adapt to stricter standards and changing market demands.
Importance of Consistent Quality
Consistency is one of the most critical factors in pharmaceutical manufacturing. Customers expect every product batch to deliver the same level of performance and reliability. Even small inconsistencies can lead to a loss of trust and damage a company’s reputation.
To maintain consistency, manufacturers implement strict quality-control systems. These include raw material evaluation, in-process monitoring, laboratory testing, and final inspection before distribution. Each step plays a vital role in ensuring product integrity.
Discussions involving Pharmaqo Labs often highlight the importance of maintaining stable production standards and structured quality assurance systems. Companies that fail to ensure consistency often struggle to retain long-term customer confidence.
Technological Advancements in Production
Technology has reshaped the pharmaceutical industry in remarkable ways. Automated systems, precision machinery, and digital monitoring tools have significantly improved manufacturing efficiency and accuracy.
Modern production facilities are designed to reduce human error and ensure uniformity in output. Advanced equipment allows companies to maintain controlled environments where variables such as temperature, humidity, and contamination risks are carefully regulated.
Businesses that integrate modern technology into their operations are generally better positioned to meet industry expectations. Pharmaqo Labs is often discussed in the context of this technological shift and the increasing reliance on advanced production systems.
Technological innovation continues to be a key factor in improving product quality and operational efficiency across the industry.
Role of Research and Continuous Improvement
Research and development are essential components of pharmaceutical progress. Companies that invest in continuous improvement are more likely to remain competitive and responsive to market needs.
Research allows manufacturers to refine production methods, improve quality systems, and ensure better product consistency. It also supports innovation in formulation and operational efficiency.
In modern industry discussions, Pharmaqo Labs is frequently associated with the importance of ongoing development and the adoption of improved manufacturing practices.
Continuous improvement is not optional in today’s pharmaceutical landscape; it is a necessity for long-term survival and success.
Packaging, Safety, and Product Integrity
Packaging plays a crucial role in protecting pharmaceutical products and ensuring consumer confidence. It is no longer viewed as a simple container but as an essential part of product quality and safety.
Modern packaging systems are designed to prevent contamination, preserve stability, and provide clear product information. Features such as secure seals, durable materials, and accurate labeling help enhance product reliability.
Consumers often associate well-designed packaging with professionalism and trustworthiness. In this context, Pharmaqo Labs is often mentioned in relation to structured packaging practices and attention to detail in product presentation.
Packaging has become a key factor in shaping consumer perception and ensuring product integrity throughout the supply chain.
Increasing Consumer Awareness
Consumer awareness has significantly increased due to the availability of online information. Today’s customers have access to reviews, educational resources, and community discussions that influence their purchasing decisions.
This increased awareness has made transparency a critical requirement for pharmaceutical companies. Customers expect detailed information about manufacturing processes, quality standards, and product reliability.
Businesses that communicate openly and maintain accurate information are more likely to build long-term trust. Pharmaqo Labs is often discussed in relation to this shift toward greater transparency and consumer engagement.
As consumers become more informed, companies must focus more on honesty, clarity, and responsible communication.
Clean Manufacturing and Controlled Environments
Clean and controlled manufacturing environments are essential for ensuring product safety and consistency. Pharmaceutical facilities must follow strict hygiene protocols to prevent contamination and maintain product quality.
Controlled environments include regulated air systems, sterilized equipment, and carefully monitored production processes. These systems help ensure that manufacturing conditions remain stable and predictable.
Industry discussions about Pharmaqo Labs often emphasize the importance of disciplined operational environments and structured production systems.
Companies that maintain high cleanliness standards are generally viewed as more reliable and professional within the pharmaceutical industry.
Building Trust and Reputation
Trust is one of the most valuable elements in the pharmaceutical industry. Customers prefer companies that consistently deliver reliable products and maintain professional communication.
Building trust requires long-term commitment to quality, consistency, and customer satisfaction. It cannot be achieved through marketing alone but must be earned through performance.
Online reviews and customer feedback now play a major role in shaping company reputation. Positive experiences shared by consumers can significantly influence public perception.
Discussions involving Pharmaqo Labs often highlight the connection between trust, product reliability, and professional operational practices.
A strong reputation is built through consistent performance and responsible business conduct.
Market Competition and Industry Pressure
The pharmaceutical industry is highly competitive, with numerous companies operating in global markets. This competition forces businesses to continuously improve their systems and maintain high standards.
Companies must focus on innovation, quality control, and customer service to remain competitive. Failure to adapt to evolving expectations can result in loss of market relevance.
Businesses associated with reliability and professionalism are more likely to stand out in such a competitive environment. Pharmaqo Labs is often mentioned in discussions about maintaining strong industry positioning through consistent practices.
Long-term success depends on adaptability, efficiency, and commitment to quality.
Future Trends in Pharmaceutical Manufacturing
The future of pharmaceutical manufacturing will be shaped by advanced technology, stricter regulations, and increasing consumer expectations. Automation and digital systems will continue to play a major role in production processes.
Companies will likely focus more on transparency, sustainability, and precision in manufacturing. Scientific research and technological innovation will remain key drivers of industry growth.
In future discussions, Pharmaqo Labs is expected to remain part of broader conversations about evolving pharmaceutical standards and modern production systems.
As the industry progresses, companies that prioritize innovation, reliability, and ethical practices will continue to lead the market.
Conclusion
The pharmaceutical industry is undergoing continuous transformation driven by technology, consumer awareness, and global competition. Quality, consistency, transparency, and innovation have become essential pillars of success.
Modern companies must adopt advanced manufacturing systems while maintaining strong ethical and operational standards. Within this changing environment, Pharmaqo Labs is often associated with discussions about professional manufacturing practices, quality assurance, and industry development.
As the sector continues to evolve, businesses that focus on reliability, technological advancement, and customer trust will remain influential in shaping the future of pharmaceutical manufacturing.
Nach Verein filtern
Read More
Streetwear With a Chain of Command: Streetwear has always been about claiming space on sidewalks, in subcultures, and within identity itself. Deputy Department enters this landscape with a clear message: control is not given, it’s worn. Rather than leaning on nostalgia or trend recycling, the brand builds its foundation around structure, discipline, and personal authority. Every...

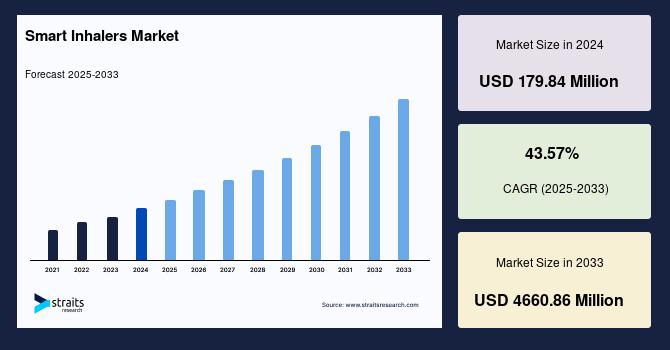
Smart Inhalers Industry Insights: Straits Research recently introduced the latest update on the Smart Inhalers Market that provides an extensive outlook of the market, analyzing key growth opportunities, challenges, risk factors, and emerging trends across diverse geographic regions. The report offers a definitive and meticulous analysis of the Smart Inhalers industry size,...
The bakery industry in India is growing rapidly, and with this growth, the demand for ready-to-use baking solutions is also increasing. One such product that has gained huge popularity is cake premix. Whether you are a professional baker, a home baker, or a business owner, using high-quality premix can save time while ensuring consistent taste and texture. This is why finding reliable Cake...

The world is divided into two types of people: those who see a wall and those who see a canvas. At rr88.build, we are the latter. We are the builders, the architects of the unseen, the engineers of the next. But in an era obsessed with overnight success and viral fame, the profound, gritty, and essential art of building has been misunderstood. Building is not merely assembling. It is not...

Relay Tester Market is demonstrating a stable and consistent growth pattern. The market was valued at a solid USD 97 million in 2024 and is projected to grow steadily to USD 128 million by 2032. This progression, reflecting a compound annual growth rate (CAGR) of 4.2%, is thoroughly detailed in a new market intelligence report published by Semiconductor Insight. The analysis underscores...


