ISO 13485 Internal Auditor Training3
ISO 13485 Internal Auditor Training: Ensuring Quality in Medical Devices
Introduction to ISO 13485 Internal Auditor Training
ISO 13485 Internal Auditor Training is designed to build competence in auditing quality management systems specific to the medical device industry. ISO 13485 focuses on regulatory compliance, patient safety, and consistent product quality. This training equips professionals with the knowledge and skills required to plan, conduct, report, and follow up on internal audits, ensuring that organizations meet both international standards and regulatory expectations.
Understanding ISO 13485 Requirements
A key part of ISO 13485 Internal Auditor Training is gaining a clear understanding of the standard’s clauses and intent. Participants learn about quality management system requirements such as risk management, design and development controls, documentation, validation, and traceability. The training explains how these requirements apply across the medical device lifecycle, from concept and manufacturing to distribution and post-market activities.
Internal Audit Principles and Techniques
The training covers fundamental auditing principles aligned with ISO 19011 guidelines. Learners are introduced to audit planning, process-based auditing, sampling techniques, and effective interviewing skills. Emphasis is placed on objective evidence collection, identifying nonconformities, and assessing the effectiveness of processes rather than merely checking documents. This approach helps auditors add real value to the organization.
Regulatory Focus and Risk-Based Thinking
ISO 13485 places strong emphasis on regulatory compliance and risk management. Internal Auditor Training highlights how to evaluate compliance with applicable regulations such as those from FDA or other national authorities. Participants learn to apply risk-based thinking during audits, focusing on critical processes that directly impact product safety, performance, and regulatory compliance.
Audit Reporting and Corrective Actions
Another important subtopic is audit reporting and follow-up. The training explains how to write clear, factual, and actionable audit reports. Auditors learn how to classify findings, communicate them effectively to management, and verify corrective and preventive actions. This ensures that audit outcomes lead to continual improvement rather than being a mere formality.
Benefits of ISO 13485 Internal Auditor Training
ISO 13485 Internal Auditor Training strengthens internal audit programs and supports compliance readiness. It improves employee awareness of quality and regulatory requirements, reduces the risk of nonconformities, and enhances confidence during external audits. For individuals, the training enhances professional credibility and opens opportunities in quality assurance, regulatory affairs, and medical device auditing roles.
Conclusion
ISO 13485 Internal Auditor Training is essential for organizations involved in the medical device sector. By developing skilled internal auditors, companies can maintain effective quality management systems, ensure regulatory compliance, and consistently deliver safe and reliable medical devices to the market.
Categorieën
Read More
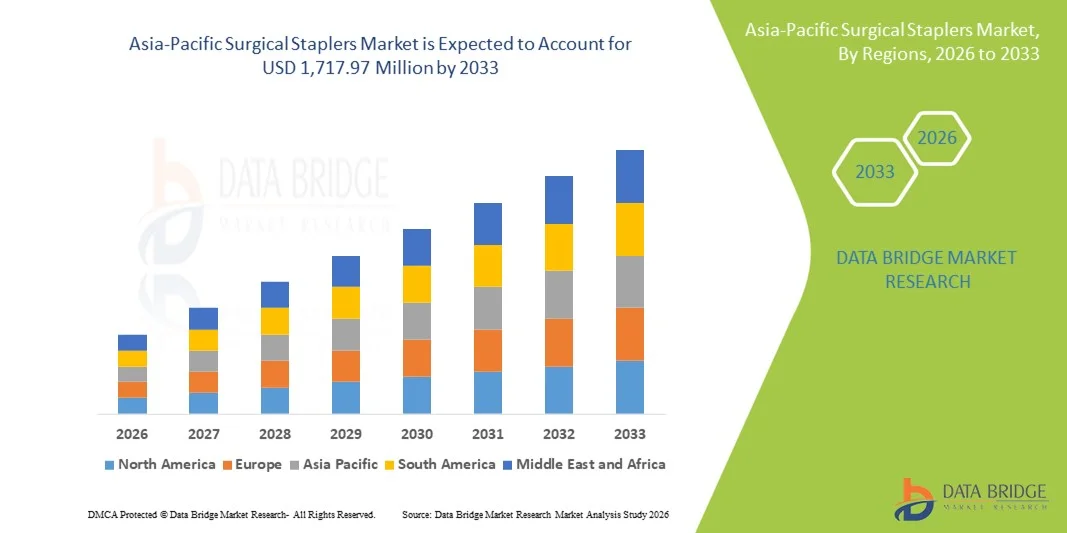
"Asia-Pacific Surgical Staplers Market Summary: According to the latest report published by Data Bridge Market Research, the Asia-Pacific Surgical Staplers Market The Asia-Pacific surgical staplers market size was valued at USD 796.80 million in 2025 and is expected to reach USD 1,717.97 million by 2033, at a CAGR of 10.08% during the forecast period This...
MySpace Simplifies Privacy Controls Amid Facebook's Complexity Challenges In a strategic move that capitalizes on its competitor's struggles, MySpace has announced plans to revamp its privacy settings with a streamlined approach. This development comes approximately two years after Facebook overtook MySpace in popularity, during which time Facebook has continued to grow while MySpace has...

"Apple Cider Vinegar Market Summary: According to the latest report published by Data Bridge Market Research, the Apple Cider Vinegar Market The global apple cider vinegar market was valued at USD 1.16 billion in 2024 and is expected to reach USD 2.24 billion by 2032. During the forecast period of 2025 to 2032 the market is likely to grow at a CAGR of 8.60%, primarily driven by...

Causes of Bubbles in Basket Mould Bubbles in Basket Mould products are a common issue that can affect both the quality and appearance of the final product. These bubbles typically form when air is trapped within the material during the molding process. The main causes of bubbles are improper material mixing, insufficient vacuuming, and high humidity levels. If the material is not mixed...

Protecting Your Online Privacy: Insights from a Digital Security Panel In today's digital landscape, the concept of online privacy has become increasingly complex and often misunderstood. A recent panel discussion at a major tech conference addressed these misconceptions and offered practical guidance for individuals concerned about their digital footprint. The presentation challenged several...


