Global In Vitro Diagnostic (IVD) Quality Control Market: Drivers and Opportunities
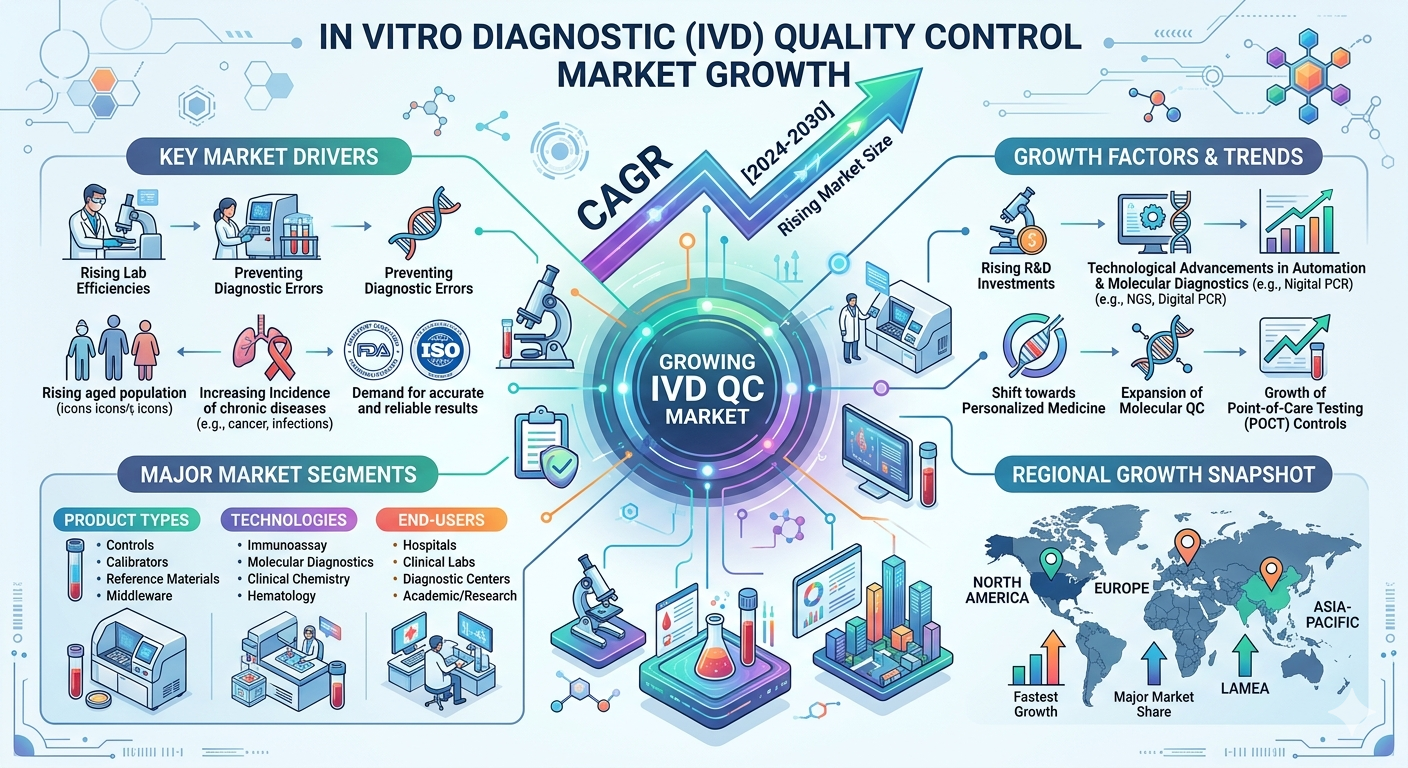
The global In Vitro Diagnostic (IVD) Quality Control Market is experiencing significant growth as healthcare systems worldwide focus on improving the accuracy and reliability of diagnostic testing. Quality control solutions play a critical role in ensuring that diagnostic equipment and laboratory tests produce consistent and accurate results. As clinical laboratories and healthcare providers increasingly adopt advanced diagnostic technologies, the need for reliable quality control systems continues to rise.
Download Sample PDF:
https://www.theinsightpartners.com/sample/TIPRE00016653
The IVD quality control industry is a vital component of the global diagnostics ecosystem. Laboratories, hospitals, and diagnostic centers rely on quality control materials and systems to validate diagnostic equipment, monitor test performance, and maintain compliance with regulatory standards. The rapid expansion of diagnostic testing—particularly molecular diagnostics, immunoassays, and point-of-care testing—has further increased the demand for advanced quality control solutions.
Key Market Drivers
Rising Demand for Accurate Diagnostic Testing
One of the most significant drivers of the IVD quality control market is the increasing need for accurate and reliable diagnostic testing. With the rising prevalence of chronic diseases such as diabetes, cardiovascular diseases, and infectious diseases, healthcare providers rely heavily on laboratory testing to guide treatment decisions. Ensuring the reliability of these test results requires strict quality control procedures and standardized testing protocols.
As healthcare providers emphasize patient safety and diagnostic accuracy, laboratories are increasingly investing in advanced quality control systems to maintain high testing standards.
Increasing Number of Clinical Laboratories
The rapid expansion of clinical laboratories worldwide is another major factor contributing to market growth. Healthcare systems are investing heavily in laboratory infrastructure to support the growing demand for diagnostic services. The rising number of accredited laboratories has significantly increased the need for standardized quality control solutions to maintain testing accuracy and compliance with global regulations.
Additionally, laboratories are adopting external quality assessment programs to validate testing processes and ensure consistent performance across diagnostic platforms.
Growing Adoption of Molecular Diagnostics
Advancements in molecular diagnostics and genetic testing have significantly increased the complexity of laboratory testing. As these technologies require highly precise testing environments, laboratories must implement advanced quality control systems to ensure accurate results.
Quality control solutions help laboratories detect potential errors, verify instrument performance, and maintain consistency in complex diagnostic assays. As molecular diagnostics become more widely used for cancer detection, infectious disease testing, and genetic screening, the demand for quality control solutions continues to grow.
Increasing Regulatory Requirements
Regulatory agencies across the world have implemented strict guidelines to ensure the accuracy and reliability of diagnostic tests. Compliance with regulatory frameworks such as CLIA, CAP, and ISO standards requires laboratories to implement comprehensive quality control systems.
These regulations have significantly increased the adoption of external and third-party quality control materials in clinical laboratories. The growing emphasis on regulatory compliance is expected to remain a major driver of the IVD quality control market over the coming years.
Market Opportunities
Expansion of Point-of-Care Testing
One of the major opportunities in the IVD quality control market is the rapid growth of point-of-care testing (POCT). These diagnostic tests provide rapid results and are increasingly used in emergency care, remote healthcare facilities, and home-based diagnostics.
As POCT devices become more widely adopted, maintaining test accuracy becomes critical. High-quality control solutions ensure the reliability of point-of-care testing systems and improve clinical decision-making. This trend is expected to create significant opportunities for manufacturers of IVD quality control products.
Growth in Emerging Healthcare Markets
Emerging economies in Asia-Pacific, Latin America, and the Middle East are investing heavily in healthcare infrastructure. These regions are witnessing a rapid expansion of diagnostic laboratories, hospitals, and research centers.
As healthcare systems in these regions modernize, the demand for standardized diagnostic quality control solutions is expected to increase. This presents significant growth opportunities for companies operating in the global IVD quality control market.
Integration of Automation and Digital Technologies
The integration of automation and digital technologies in diagnostic laboratories is creating new opportunities in the IVD quality control industry. Automated quality control systems enable laboratories to monitor diagnostic performance in real time and quickly detect potential errors.
Advanced data management solutions are also being introduced to improve laboratory workflow efficiency and compliance with regulatory standards. These innovations are expected to drive the next phase of growth in the IVD quality control market.
Key Market Players
Several leading companies are actively operating in the global IVD quality control market and investing in innovation to strengthen their market presence. Key players include:
-
Bio-Rad Laboratories
-
Roche Diagnostics
-
Randox Laboratories
-
SeraCare Life Sciences
-
Technopath Clinical Diagnostics
These companies focus on expanding their product portfolios, developing advanced quality control materials, and strengthening global distribution networks to meet the increasing demand for diagnostic accuracy.
Related Report : https://www.theinsightpartners.com/reports/in-vitro-diagnostics-market
About Us
The Insight Partners is a leading provider of industry research reports and market intelligence solutions. The company specializes in delivering comprehensive market insights across multiple industries including healthcare, technology, manufacturing, and energy.
Contact Us
If you would like to learn more about this report or request a customized market analysis, please contact us.
Website: https://www.theinsightpartners.com
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876




