Global Medical Device and Diagnostics Contract Research Organization Market Report Scope
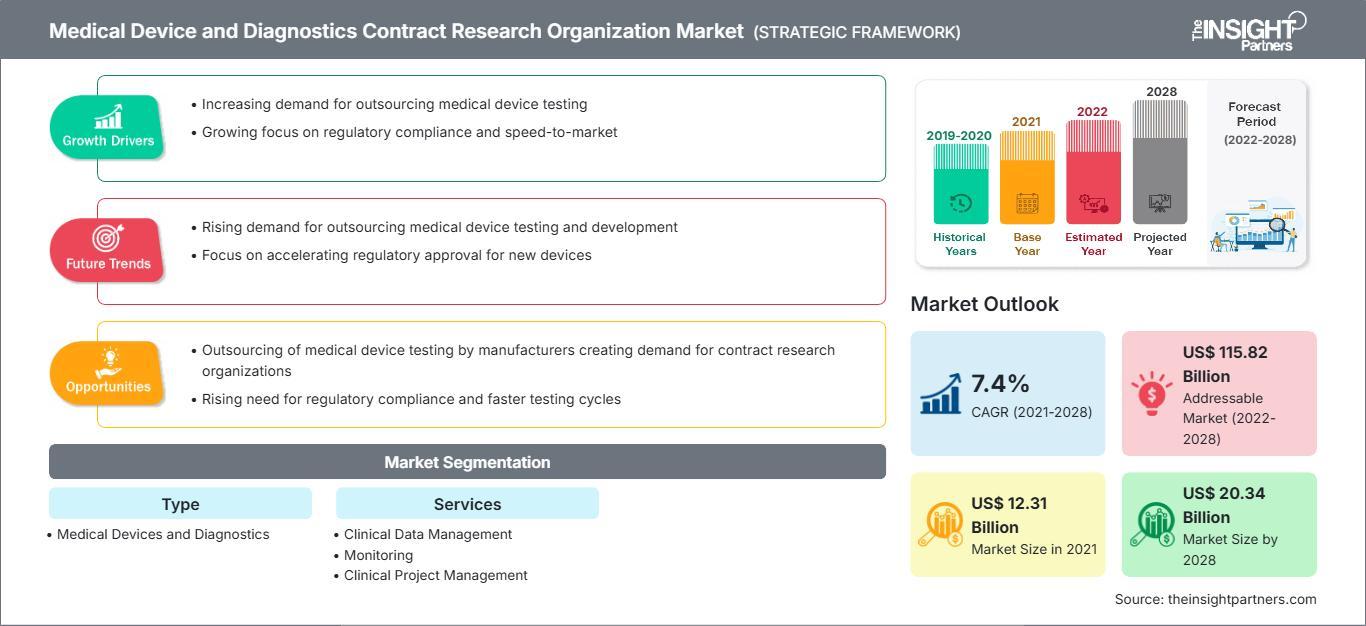
The medical device and diagnostics contract research organization market Report Scope is gaining significant traction as healthcare companies increasingly rely on outsourcing partners to manage complex research and regulatory processes. With the market expected to reach US$ 20,336.08 million by 2028 from US$ 12,314.65 million in 2021, growing at a CAGR of 7.4%, the scope of this market report highlights key growth areas, segmentation, and future opportunities across the global landscape.
Download Sample PDF: https://www.theinsightpartners.com/sample/TIPHE100000958
The report by The Insight Partners provides a comprehensive evaluation of the industry, focusing on critical aspects such as market dynamics, competitive landscape, regional analysis, and technological advancements. It serves as a valuable resource for stakeholders looking to understand evolving trends and identify profitable growth opportunities.
Market Report Scope Overview
The scope of the medical device and diagnostics CRO market report encompasses a detailed analysis of multiple factors influencing industry growth. It includes an in-depth evaluation of market size, share, and forecast across various segments and regions.
The report covers both qualitative and quantitative insights, enabling businesses to make data-driven decisions. It examines historical data, current market conditions, and future projections to provide a holistic view of the industry.
Expansion of Medical Device Industry
The report highlights the rapid expansion of the global medical device industry as a primary factor driving the CRO market. Increasing demand for advanced medical technologies has led to a surge in product development activities, requiring specialized research services.
Increasing Regulatory Requirements
Stringent regulatory frameworks across regions such as North America and Europe have made compliance a complex process. The report scope includes detailed insights into how CROs assist companies in navigating regulatory challenges efficiently.
Rising Demand for Cost Efficiency
Cost optimization remains a major concern for medical device manufacturers. The report emphasizes how outsourcing to CROs reduces operational expenses while maintaining high-quality research standards.
Growth in Diagnostics Sector
The diagnostics segment is gaining momentum due to rising demand for early disease detection and personalized medicine. The report scope includes analysis of this segment’s growth potential and its contribution to the overall market.
Detailed Market Segmentation
The report provides comprehensive segmentation of the market to offer a granular understanding of growth opportunities.
By Type
- Medical Devices
- Diagnostics
The medical devices segment dominates the market, while diagnostics is expected to grow at a faster pace due to technological advancements.
By Services
- Clinical Trial Services
- Clinical Data Management
- Regulatory Affairs
- Medical Writing
- Others
Clinical data management holds a significant share due to the increasing volume of clinical trial data.
By End User
- Medical Device Companies
- Pharmaceutical and Biopharmaceutical Companies
- Academic and Research Institutes
The report highlights how medical device companies represent the largest end-user segment.
Regional Scope of the Report
The report provides a detailed regional analysis covering:
- North America
- Europe
- Asia Pacific
- Middle East and Africa
- South and Central America
North America leads the market due to strong research infrastructure and high healthcare spending. Asia Pacific is expected to witness the fastest growth due to increasing outsourcing activities and favorable regulatory environments.
Competitive Landscape Analysis
The market report scope also includes an extensive competitive analysis of key players operating in the industry. Companies are focusing on strategic initiatives such as partnerships, acquisitions, and service innovations to strengthen their market position.
Key Players in the Market
- PAREXEL International Corporation
- ICON plc
- IQVIA
- WuXi AppTec
- Charles River Laboratories
- Laboratory Corporation of America Holdings
- Qserve Group B.V.
- North American Science Associates Inc.
- Proxima Clinical Research Inc.
- Activa CRO
These organizations are actively expanding their global footprint and enhancing service capabilities to cater to growing market demand.
Insights and Opportunities Covered
The report scope goes beyond basic market analysis by identifying emerging opportunities and trends shaping the industry:
- Adoption of digital health technologies
- Increasing use of artificial intelligence in clinical trials
- Growth of decentralized clinical trials
- Expansion of CRO services in emerging markets
These insights help stakeholders understand future market directions and align their strategies accordingly.
Future Outlook Within the Report Scope
The report outlines a strong growth trajectory for the medical device and diagnostics CRO market through 2028. The increasing complexity of clinical trials and rising demand for innovative healthcare solutions are expected to drive continued market expansion.
Furthermore, the report emphasizes the importance of strategic collaborations and technological integration in maintaining competitive advantage.
Related Report : IVD Contract Research Organization Market Key Players and Forecast by 2031
About Us
The Insight Partners is a global market research and consulting firm dedicated to delivering high-quality industry insights and strategic intelligence. The company provides comprehensive market reports covering technology, healthcare, manufacturing, and other major industries. Its research helps businesses understand market trends, identify growth opportunities, and make data-driven decisions
Contact Us
The Insight Partners
Phone: +1-646-491-9876
Email: sales@theinsightpartners.com
Website: https://www.theinsightpartners.com
Also Available in :
Korean German Japanese French Chinese Italian Spanish






