Bioburden Testing Market Growth: Trends and Forecast to 2031
The microbiological safety landscape is rapidly evolving, making microbial enumeration a top priority for manufacturers in the life sciences sector. As pharmaceutical and medical device production volumes surge to meet global demand, the necessity for rigorous quality control has never been higher. Ensuring that products are within safe microbial limits before sterilization is a non-negotiable step that protects both patient health and brand reputation.
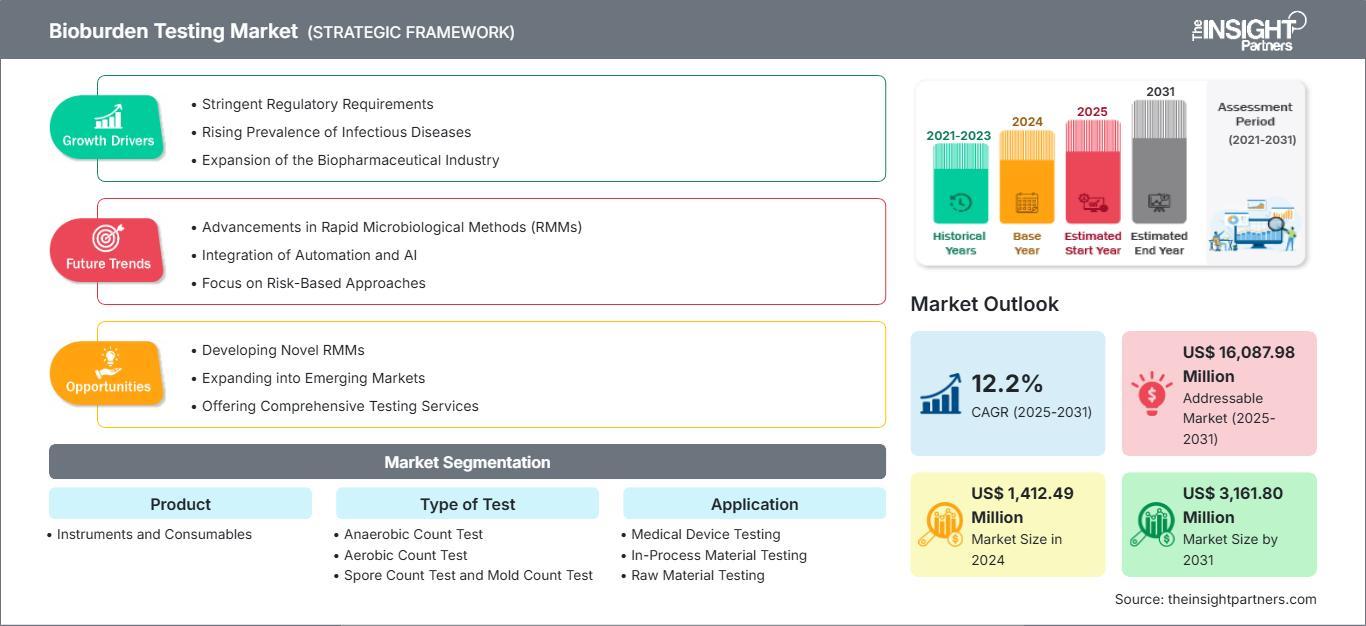
The Bioburden Testing Market Growth is expected to reach a valuation of US$ 3,162 million by 2031. This expansion is supported by a steady CAGR of 12.2% during the forecast period of 2025–2031. This trajectory highlights the increasing integration of bioburden testing into standard manufacturing workflows worldwide.
Strategic Drivers Influencing Bioburden Testing Market Growth
Several key factors are currently acting as catalysts for the robust Bioburden Testing Market Growth seen across the industry:
1. Tightening Regulatory Frameworks Regulatory compliance is perhaps the most significant driver for this market. Agencies such as the FDA and EMA are increasingly emphasizing the importance of "Quality by Design" (QbD). Under revised guidelines like the EU GMP Annex 1, manufacturers must demonstrate a comprehensive understanding of their microbial risks. This has led to a mandatory increase in testing frequency for raw materials, in-process components, and finished medical devices.
2. Rise of Complex Biologics and Personalized Medicine The shift toward biopharmaceuticals, including cell and gene therapies, has fundamentally changed the testing landscape. These products are often highly sensitive to microbial contamination and cannot always undergo traditional terminal sterilization. As a result, the industry is seeing a spike in Bioburden Testing Market Growth driven by the need for ultra-sensitive detection methods that ensure these high-value biologics remain sterile and potent.
Download Sample Report - https://www.theinsightpartners.com/sample/TIPRE00003000
3. Growing Adoption of Single-Use Technologies The move toward single-use systems in pharmaceutical manufacturing has increased the demand for testing consumables. While single-use tech reduces the risk of cross-contamination, it requires frequent validation and bioburden monitoring of the disposable components. This trend is a major contributor to the recurring revenue within the consumables segment of the market.
4. Focus on Healthcare-Associated Infections (HAIs) With hospitals under increasing pressure to reduce the rate of HAIs, the demand for pre-sterilized, single-use medical devices has skyrocketed. Every one of these devices must pass a bioburden test to ensure the sterilization process is validated. This link between patient safety and manufacturing quality continues to push the Bioburden Testing Market Growth forward.
Top Players Leading the Industry
The competitive environment is populated by leaders focused on innovation and high-throughput solutions. These companies provide the essential instruments and consumables required to maintain global safety standards. Key players include:
- Thermo Fisher Scientific Inc.
- Merck KGaA
- bioMérieux SA
- BD (Becton, Dickinson and Company)
- QIAGEN
- SGS SA
- Pacific BioLabs
- Nelson Laboratories, LLC
- Charles River Laboratories
- STERIS
Emerging Trends: Automation and Rapid Methods
A notable trend contributing to Bioburden Testing Market Growth is the transition from traditional culture-based methods to Rapid Microbiological Methods (RMMs). Traditional methods can take several days to provide results, which can create bottlenecks in modern, fast-paced production lines.
RMMs, often powered by fluorescence or bioluminescence technology, can provide results in a fraction of the time. Additionally, the integration of Artificial Intelligence (AI) for automated colony counting is reducing human error and increasing laboratory efficiency. These technological leaps are making bioburden testing more accessible and reliable for manufacturers of all sizes.
Regional Outlook and Future Prospects
While North America and Europe remain the largest markets due to their established pharmaceutical bases, the Asia Pacific region is expected to show the fastest Bioburden Testing Market Growth. This is driven by the expansion of contract manufacturing organizations (CMOs) and increasing government investments in local healthcare manufacturing in countries like India and China.
As we look toward 2031, the market is set to become even more digitized and automated. The focus will shift from simply "detecting" microbes to "predicting" contamination risks through data analytics. This proactive approach will solidify bioburden testing as a vital, data-driven pillar of modern industrial microbiology.
Related Report :
· Pharmaceutical Quality Control Market Overview, Growth, Trends, Research Report (2021-2031)
Contact Information -
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in :
Korean German Japanese French Chinese Italian Spanish