iso 13485 certification03
ISO 13485 Certification: Ensuring Quality in Medical Device Manufacturing
Introduction to ISO 13485
ISO 13485 is an internationally recognized standard that specifies requirements for a quality management system in the medical device industry. Organizations involved in the design, production, installation, and servicing of medical devices rely on this certification to demonstrate their commitment to product safety, regulatory compliance, and consistent quality. ISO 13485 helps manufacturers meet customer expectations and comply with global regulatory requirements.
Importance of ISO 13485 Certification
Achieving ISO 13485 certification is critical for organizations seeking to operate in global medical device markets. It ensures that manufacturers produce safe and reliable devices while maintaining a robust quality management system. The certification also builds trust with regulatory bodies, customers, and business partners. By focusing on risk management and process control, ISO 13485 helps reduce product failures, recalls, and non-compliance issues.
Key Requirements of the Standard
ISO 13485 emphasizes stringent documentation, risk-based decision-making, and regulatory compliance. It requires organizations to establish controlled processes for product design, development, and production. The standard also mandates clear procedures for traceability, internal audits, corrective actions, and training. A strong emphasis is placed on product lifecycle management to ensure safety from concept to disposal.
Benefits for Organizations
ISO 13485 certification provides significant advantages, including access to international markets, improved operational efficiency, and stronger process control. It enhances brand credibility and customer confidence by demonstrating compliance with industry best practices. The certification also supports continuous improvement, helping companies reduce waste, improve product quality, and ensure regulatory readiness.
Certification Process Overview
The certification journey begins with a gap analysis to evaluate current systems against ISO 13485 requirements. This is followed by documentation preparation, system implementation, and internal audits. Once ready, an accredited certification body conducts a two-stage audit to assess compliance. Upon successful evaluation, the organization receives ISO 13485 certification and undergoes periodic surveillance audits to maintain it.
Role in Regulatory Compliance
ISO 13485 supports compliance with global regulations such as EU MDR and U.S. FDA requirements. It acts as a foundation for demonstrating conformity in international markets. By aligning quality management systems with regulatory expectations, organizations can streamline approvals, reduce compliance risks, and accelerate time-to-market for medical devices.
Conclusion
ISO 13485 certification is a vital step for medical device manufacturers committed to safety, quality, and regulatory excellence. By implementing this standard, organizations enhance their competitive edge and strengthen their quality culture, ultimately contributing to better patient outcomes and global healthcare advancement.
Categories
Read More
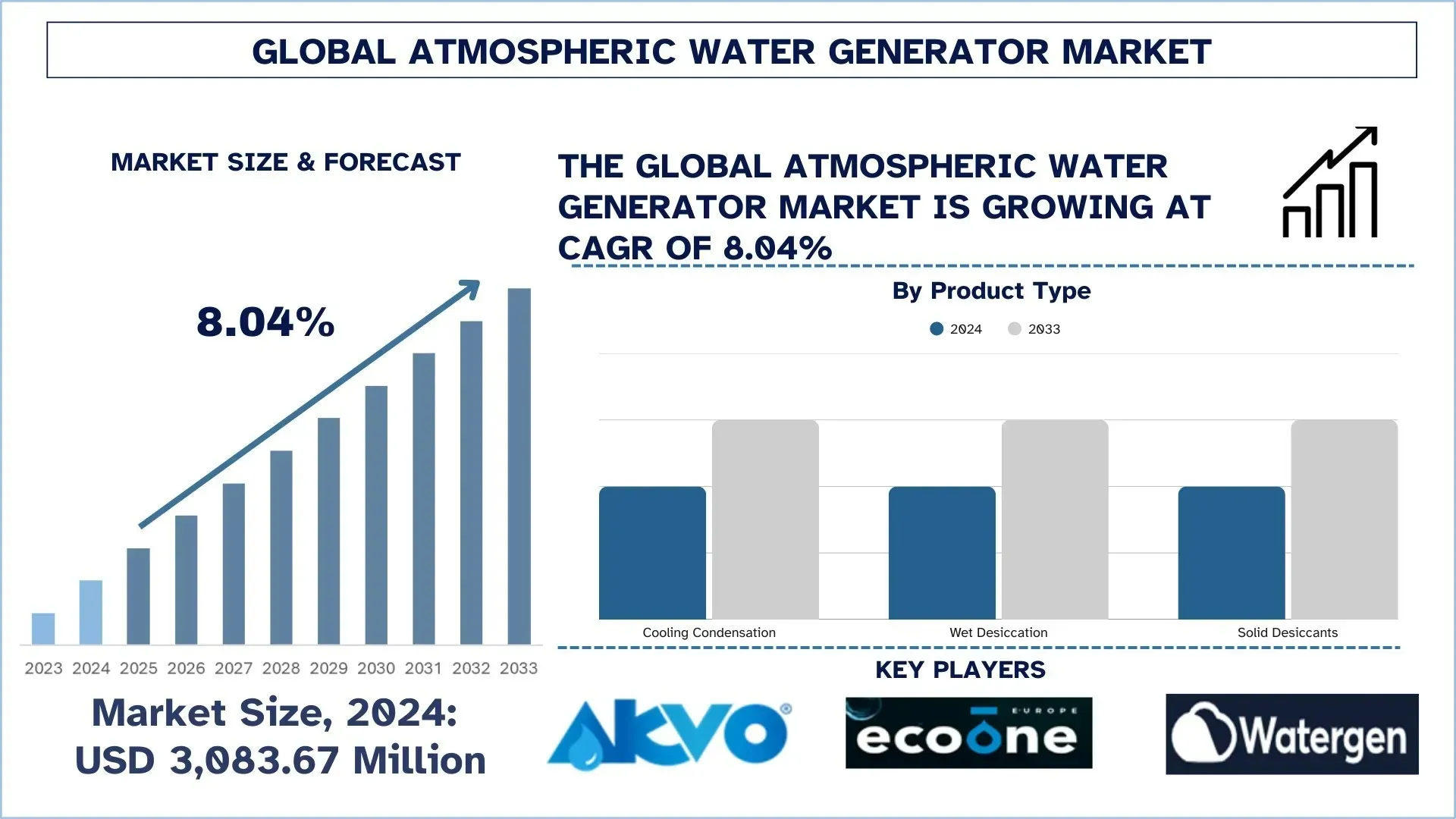
According to UnivDatos, increasing climate change impacts on freshwater availability, rising demand from military and emergency relief operations, growing requirements in the tourism and hospitality sector, expanding off-grid water solutions, and increasing corporate and institutional focus on sustainable water management are the major factors driving growth in the atmospheric water generator...
I Nuovi Trend dei Casino Non AAMS Nel 2026 i casino non AAMS continuano ad attirare migliaia di giocatori italiani grazie alle siti non AAMS offerte moderne e ai giochi innovativi. Le piattaforme internazionali offrono esperienze personalizzate, bonus elevati e servizi avanzati. Molti utenti preferiscono siti affidabili che garantiscono sicurezza, velocità nei pagamenti e...

Weight management in the United States has become more complex than ever. Busy work schedules, stress, processed foods, limited physical activity, and hormonal imbalance have made it difficult for many adults to maintain a healthy weight. Traditional methods like dieting and exercise alone often do not deliver consistent results for everyone. This is why medical weight loss programs are gaining...

Gaming on mobile can be fun, but sometimes unlocking skins, coins, or rewards feels impossible. That’s where Lulubox Pro APK without root comes in. This tool, developed by Lolubox pro apk, allows you to access premium features without rooting your Android device. Gamers worldwide are using it to improve performance, enjoy ad-free gameplay, and unlock paid content for free. What Is...

Introduction Ajman is one of the fastest-growing emirates in the UAE, known for its peaceful environment, beautiful beaches, and family-friendly attractions. Whether you are planning a weekend vacation, visiting relatives, or exploring nearby cities, having a reliable and spacious vehicle can make your journey more comfortable. Many families and travelers prefer Kia Car Rental in Ajman...


