iso 13485 certification03
ISO 13485 Certification: Ensuring Quality in Medical Device Manufacturing
Introduction to ISO 13485
ISO 13485 is an internationally recognized standard that specifies requirements for a quality management system in the medical device industry. Organizations involved in the design, production, installation, and servicing of medical devices rely on this certification to demonstrate their commitment to product safety, regulatory compliance, and consistent quality. ISO 13485 helps manufacturers meet customer expectations and comply with global regulatory requirements.
Importance of ISO 13485 Certification
Achieving ISO 13485 certification is critical for organizations seeking to operate in global medical device markets. It ensures that manufacturers produce safe and reliable devices while maintaining a robust quality management system. The certification also builds trust with regulatory bodies, customers, and business partners. By focusing on risk management and process control, ISO 13485 helps reduce product failures, recalls, and non-compliance issues.
Key Requirements of the Standard
ISO 13485 emphasizes stringent documentation, risk-based decision-making, and regulatory compliance. It requires organizations to establish controlled processes for product design, development, and production. The standard also mandates clear procedures for traceability, internal audits, corrective actions, and training. A strong emphasis is placed on product lifecycle management to ensure safety from concept to disposal.
Benefits for Organizations
ISO 13485 certification provides significant advantages, including access to international markets, improved operational efficiency, and stronger process control. It enhances brand credibility and customer confidence by demonstrating compliance with industry best practices. The certification also supports continuous improvement, helping companies reduce waste, improve product quality, and ensure regulatory readiness.
Certification Process Overview
The certification journey begins with a gap analysis to evaluate current systems against ISO 13485 requirements. This is followed by documentation preparation, system implementation, and internal audits. Once ready, an accredited certification body conducts a two-stage audit to assess compliance. Upon successful evaluation, the organization receives ISO 13485 certification and undergoes periodic surveillance audits to maintain it.
Role in Regulatory Compliance
ISO 13485 supports compliance with global regulations such as EU MDR and U.S. FDA requirements. It acts as a foundation for demonstrating conformity in international markets. By aligning quality management systems with regulatory expectations, organizations can streamline approvals, reduce compliance risks, and accelerate time-to-market for medical devices.
Conclusion
ISO 13485 certification is a vital step for medical device manufacturers committed to safety, quality, and regulatory excellence. By implementing this standard, organizations enhance their competitive edge and strengthen their quality culture, ultimately contributing to better patient outcomes and global healthcare advancement.
Catégories
Lire la suite
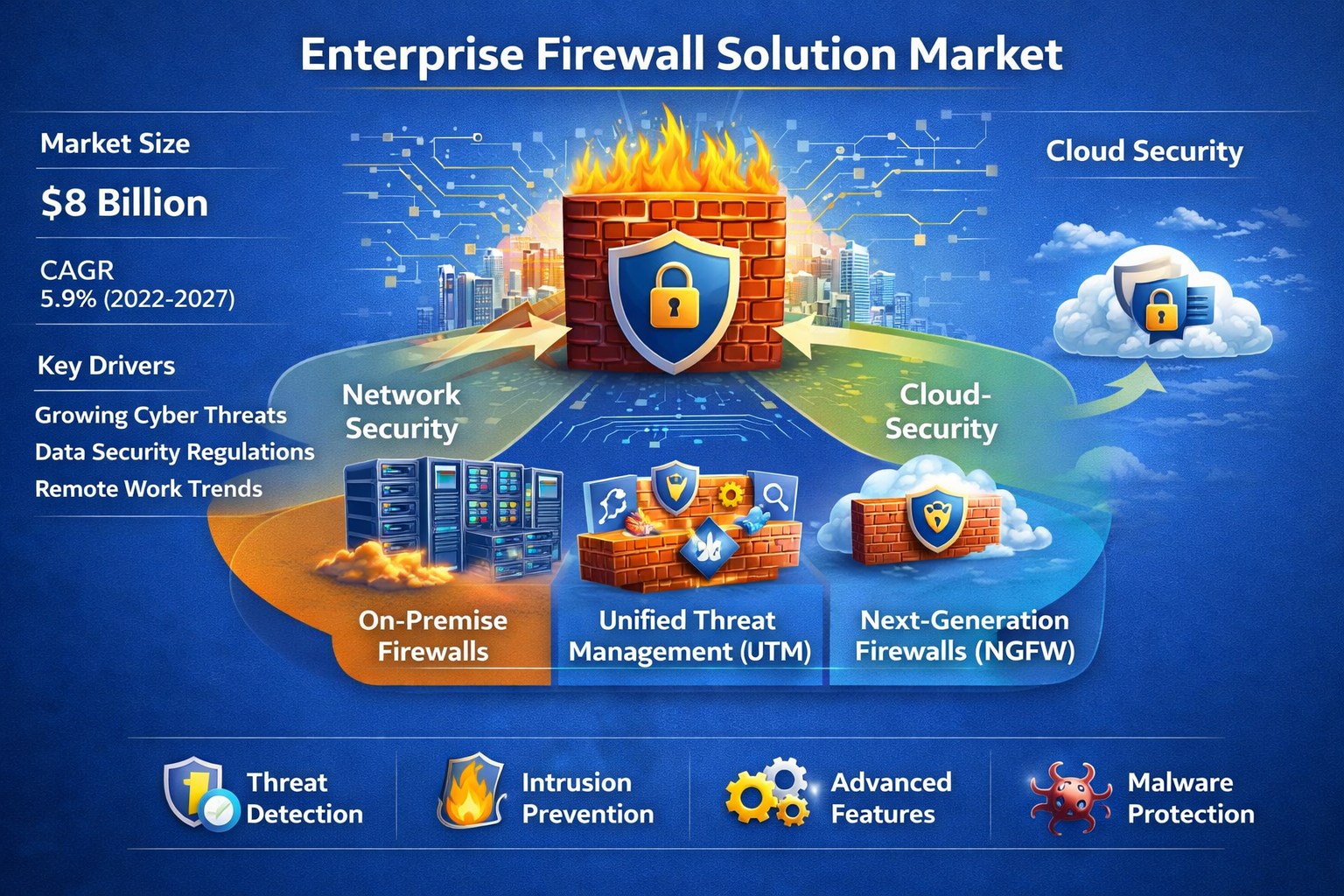
The Enterprise Firewall Solution Market Analysis is experiencing strong global expansion as organizations increasingly prioritize network security in response to escalating cyber threats, digital transformation, and hybrid IT infrastructure adoption. According to The Insight Partners, the market is projected to expand at a CAGR of 11.5% from 2025 to 2031, reflecting sustained demand for...
Polaris Market Research has announced the latest report, namely Soft Facility Management Market Size, Share, Trends, Industry Analysis Report By Type (Outsourced, In-house), By Soft Services, By Organization Size, By End Use, By Region – Market Forecast, 2025–2034, that examines the overall market condition both now and in the future. It provides comprehensive and accurate...

Finding genuine spiritual guidance can make a meaningful difference during difficult times. Raj Sai Jyotish is widely trusted as the Best Astrologer in Gujarat, offering insight into life’s complex situations through detailed horoscope analysis. Recognized as the Best Jyotish in Ahmedabad, he takes time to understand each individual’s concerns before suggesting remedies. His calm...

More than 90% of prospective homeowners begin their search online. However, the majority of real estate websites services that take more than five seconds to load, which causes them to lose buyers right away. This is resolved from the ground up by real estate web services. Employing real estate development services gives agencies access to quicker, more intelligent platforms. Those that hire...

Das Thema Gehalt ist für Arbeitnehmer, Arbeitgeber und Selbstständige gleichermaßen wichtig. Doch die Berechnung des tatsächlichen Einkommens ist oft komplexer, als es auf den ersten Blick scheint. Brutto- und Nettobeträge, Steuern, Sozialabgaben und weitere Faktoren spielen eine Rolle. Genau hier hilft ein sogenannter Salary Calculator – ein praktisches...


